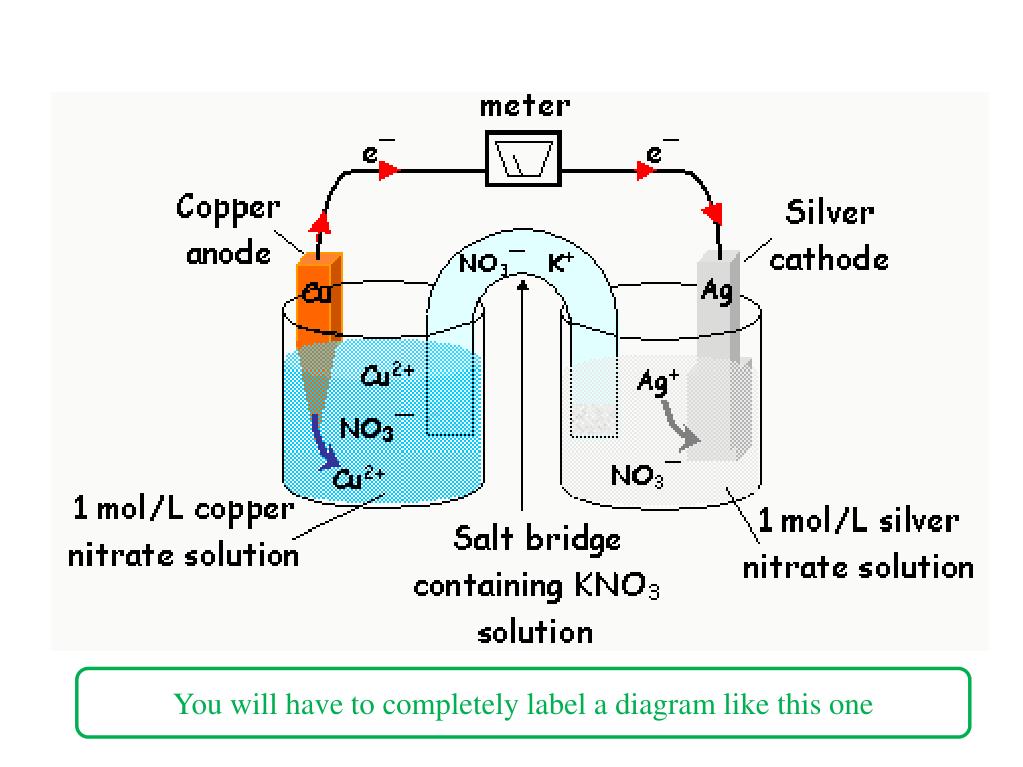
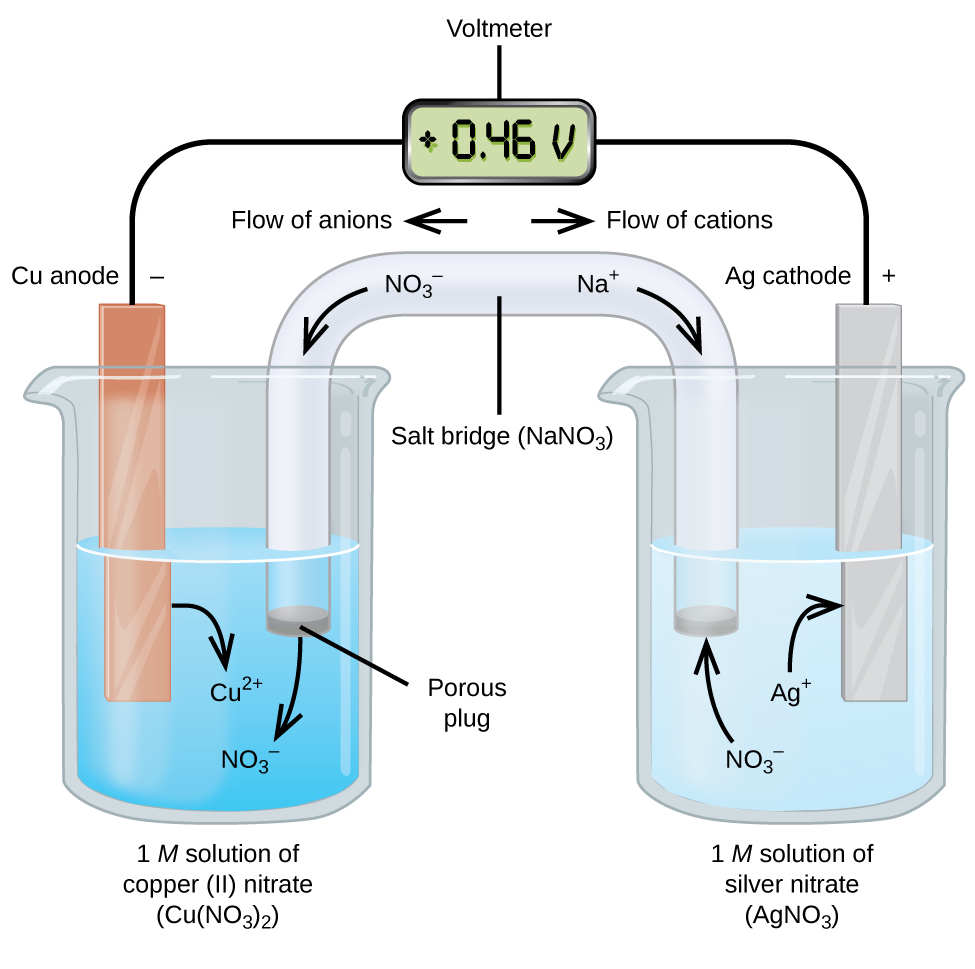
 The metal's differences in oxidation/reduction potential drive the reaction until equilibrium. A salt bridge or porous membrane connects the two solutions, keeping electric neutrality and the avoidance of charge accumulation. Each metal is in a separate solution often the aqueous sulphate or nitrate forms of the metal, however more generally metal salts and water which conduct current. Galvanic cell with no cation flowĪ wire connects two different metals (ex. Electrolytic cells are used in electrowinning processes.A galvanic cell (voltaic cell) named after Luigi Galvani ( Alessandro Volta) is an electrochemical cell that generates electrical energy from spontaneous redox reactions.Electrolysis is used for the electrorefining of non-ferrous metals.Electrolytic cells are used for electroplating, a process by which a thin protective layer of one metal is formed over another metal, mostly for protection. 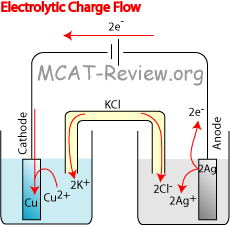
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
